Small Patients, Big Changes: The First-Ever Pediatric Neuromuscular Block Guidelines Are Here
2025 ESAIC/ESPA Pediatric NMB Guidelines: What Clinicians Need to Know—and How Modern Monitoring Meets the Moment
For the first time in the history of pediatric anesthesia, the European Society of Anaesthesiology and Intensive Care (ESAIC) and the European Society of Pediatric Anaesthesiology (ESPA) have issued dedicated clinical guidelines on neuromuscular block (NMB) in anaesthetized children. Published in the European Journal of Anaesthesiology (Veyckemans et al., 2026), this landmark document fills a critical gap — previous adult-focused guidelines offered only an informal call for pediatric-specific guidance. That call has now been answered.
The gap between what we know and what we practice has real consequences for pediatric patients. Data from the APRICOT study, cited throughout the new Pediatric Guidelines, found that only 16% of children who received a neuromuscular blocking agent (NMBA) had any neuromuscular monitoring. Absence of monitoring has been directly linked to higher rates of residual block and postoperative respiratory complications.
These new Pediatric Guidelines make the path forward clear.
EMG Over AMG: A Meaningful Upgrade for Pediatric Practice
One of the most clinically significant recommendations in the new Pediatric Guidelines is the preference for electromyography (EMG)-based monitoring over acceleromyography (AMG). The rationale is practical and well-supported: AMG devices are difficult to secure on small hands, tend to be bulky, and require calibration that can be technically challenging in neonates and infants—barriers that have historically contributed to the underuse of monitoring in the youngest patients.
EMG-based devices avoid the "staircase phenomenon" (twitch potentiation) seen with AMG, require lower stimulating current, and are now available in sizes suitable for pediatric patients. The new Pediatric Guidelines specifically note that novel EMG-based monitors have "expanded the possible clinical applications of accurate Train of Four (“TOF”) monitoring in children, including those less than 6 months of age."
This shift toward EMG is not just a technical preference—it's a recognition that monitoring tools must actually work in the patients they are intended for.
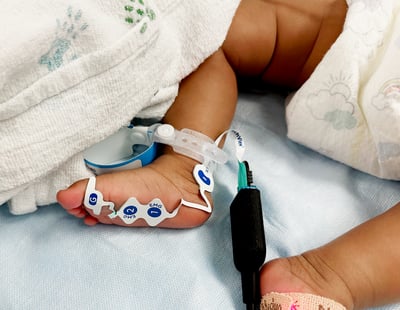
Adductor Pollicis and Flexor Hallucis Brevis: Matching the Right Tool to the Right Site
The new Pediatric Guidelines recommend the adductor pollicis as the primary monitoring site, with the flexor hallucis brevis as an alternative when upper extremity access is limited—a common scenario in pediatric cases involving complex positioning or surgical access. Research using EMG-based monitoring has confirmed meaningful differences in NMB onset and recovery timing between these two sites, reinforcing the clinical importance of site-specific, reliable electrode placement.
Meeting clinicians where they practice, the TwitchView Train of Four Monitor—an electromyography (EMG)-based neuromuscular monitor—offers three electrode sizes specifically sized for the anatomical demands of pediatric and adult patients, with configurations suited to both the adductor pollicis and flexor hallucis brevis. Already in use at 5 of the top 10 U.S. Children's Hospitals, TwitchView reflects a broader clinical recognition that pediatric monitoring requires pediatric-specific solutions. Where legacy AMG tools created real-world barriers in small patients, purpose-built EMG technology removes them.
Sugammadex Over Neostigmine—With Monitoring to Guide Every Step
The guidelines carry a strong recommendation for sugammadex over neostigmine for reversal of aminosteroid NMBAs in children, citing faster recovery, shorter time to extubation, and more reliable reversal at moderate and deep block levels. This applies to children under two years of age at the same weight-based dosing used in older patients, though the drug remains off-label in this age group at time of publication.
Importantly, sugammadex does not reduce the need for monitoring—it requires it. Appropriate dosing depends directly on quantitative TOF data: 2 mg/kg when TOF count ≥ 2, and 4 mg/kg when post-tetanic count ≥ 1. Dosing without that information is dosing without a target.
This principle extends to succinylcholine as well. Given its well-documented variability in duration—and the risk of prolonged block in patients with pseudocholinesterase deficiency or following repeated doses—the new Pediatric Guidelines recommend using both clinical assessment and quantitative neuromuscular monitoring to confirm full recovery.
Children under two years who receive large total doses of rocuronium or vecuronium warrant extended PACU observation after sugammadex reversal, given documented cases of recurarisation, the return of neuromuscular blockade and muscle weakness after an initial, often temporary, recovery to a TOF ratio of 90% or greater. Here again, quantitative monitoring is the difference between assumption and confirmed reversal.
Implementation: From Guidelines to Practice
The authors are candid that publishing guidelines is the easier part. Widespread adoption of quantitative monitoring requires education, institutional support, and tools that are genuinely usable in pediatric patients. The guidelines propose a stepwise framework—from education and qualitative monitoring to routine quantitative EMG-based monitoring as a standard of care.
The technology to support that final step is no longer aspirational. For teams ready to align with the 2025 ESAIC/ESPA recommendations, purpose-built pediatric EMG monitoring, such as TwitchView monitoring, is available today.
Learn more about how TwitchView supports guideline compliant neuromuscular monitoring across the full pediatric population.